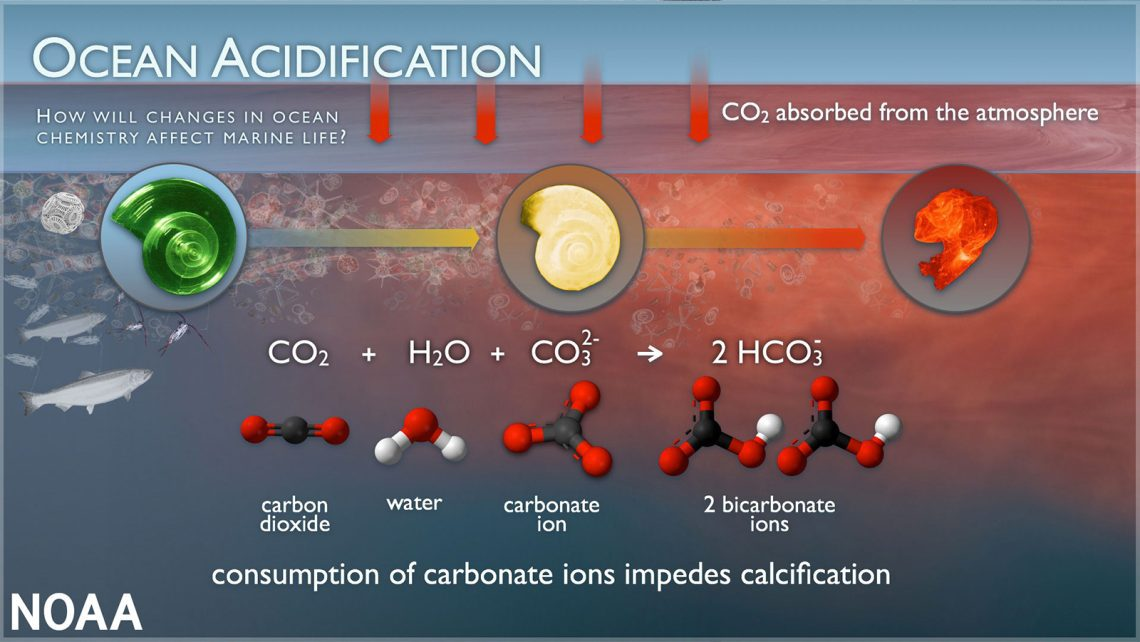
For my unessay project, I’ve decided to focus on ocean acidification, calcium carbonate deficiency in the ocean, and effects on calcifying marine organisms. Ocean acidification is the process by which the ocean’s pH declines (acidifies) through an increase in CO2 absorption. Increases in atmospheric CO2 from human activity have subsequently increased CO2 concentrations in seawater. Water and CO2 combine to form carbonic acid (H2CO3–), which then breaks down into hydrogen (H+) and bicarbonate HCO3–). This increase in H+ ions decreases the pH of the ocean. As the ocean continues to absorb CO2, the pH will continue to decrease and the ocean will continue to become more acidic.

Another consequence of ocean acidification is its impact on calcifying marine organisms. Carbonate ions (CO32-) in the water bond with excess hydrogen, which results in fewer carbonate ions available for calcifying organisms. As you can imagine, increasing levels of atmospheric (and therefore oceanic) CO2 and continued acidification will result in lower and lower concentrations of available carbonate in the water column. Organisms that require carbonate will have a more difficult time building and maintaining the structures (such as shells or skeletons) that they need to survive.
As I begin my research of my unessay, I hope to focus on a wide range of organisms to see how acidification may have varying effects. I’m hoping to begin with this simple variation and, if time and other factors permit, extend my research further to look at variation in other areas such as resistance, differences in effects due to location, adaptations, etc. For today, I’ll be doing a brief overview on how ocean acidification has affected corals, and bivalves.

Studies have shown that at atmospheric CO2 levels of 560 ppm, coral calcification could decrease by 40% due to the coral’s restricted ability to form skeletons. As of 2019, current levels are at around 410 ppm, but are only continuing to increase. It has also been shown that reduced pH/acidification can escalate calcium carbonate dissolution rates. Decreased calcification in corals reduces growth, which hinder’s the coral’s ability to compete for space against other non-calcifying organisms such as macroalgae. There is variation regarding the rates of decreased calcification in response to ocean acidification, and it is believed that other factors including temperature, light, and salinity also play a role in calcification and growth. This variation is an example of what I hope to investigate further in my unessay.

Long term exposure to decreased carbonate and pH has been shown to impact bivalves’ biological processes, ultimately leading to slower growth and reduced metabolism, particularly in mussels. Ocean acidification can also lead to shell dissolution (breakdown and decomposition) and has been observed in bivalves as well as other marine organisms. Degraded shells in bivalves will likely lead to lowered fitness rates due to reduced lifespans and higher susceptibility to predation.
Sources:
https://www.noaa.gov/education/resource-collections/ocean-coasts/ocean-acidification https://ask.ifas.ufl.edu/publication/FA220
Leave a Reply